메가슬롯
아시아 최고의 슬롯 전문 웹사이트를 경험하세요!
신규 가입 20% 보너스 + 1만 쿠폰 지급!
올인 마지막 입금액의 10% 쿠폰
레벨별 콤프 0.1%~0.6% 가입 동시 적립!
메가슬롯 홈페이지

페이지 상단에 제공된 배너나 바로가기 버튼을 사용하면 메가슬롯 공식 홈페이지 도메인으로 바로 연결됩니다.
홈페이지 좌측 상단 이벤트안내 탭을 클릭하면 현재 제공하는 이벤트를 한눈에 볼 수 있습니다.
그밖에 실시간으로 잭팟 금액 확인, 입출금 처리, 쿠폰 발급 상황 확인, 그리고 머니 이체 등 다양한 기능을 활용할 수 있습니다.
웹사이트 바닥에는 텔레그램 및 위쳇을 이용한 고객센터 연락처가 제시되어 있어, 문제가 생길 시 이 채널들을 통해 문의 가능합니다.
메가슬롯 도메인 게임 목록
메가슬롯의 공식 웹사이트 하단에서는 40개 이상의 슬롯 게임 제공업체 목록을 확인할 수 있으며, 이들은 모두 메가슬롯과 정식 계약을 맺은 정품 게임사들입니다.
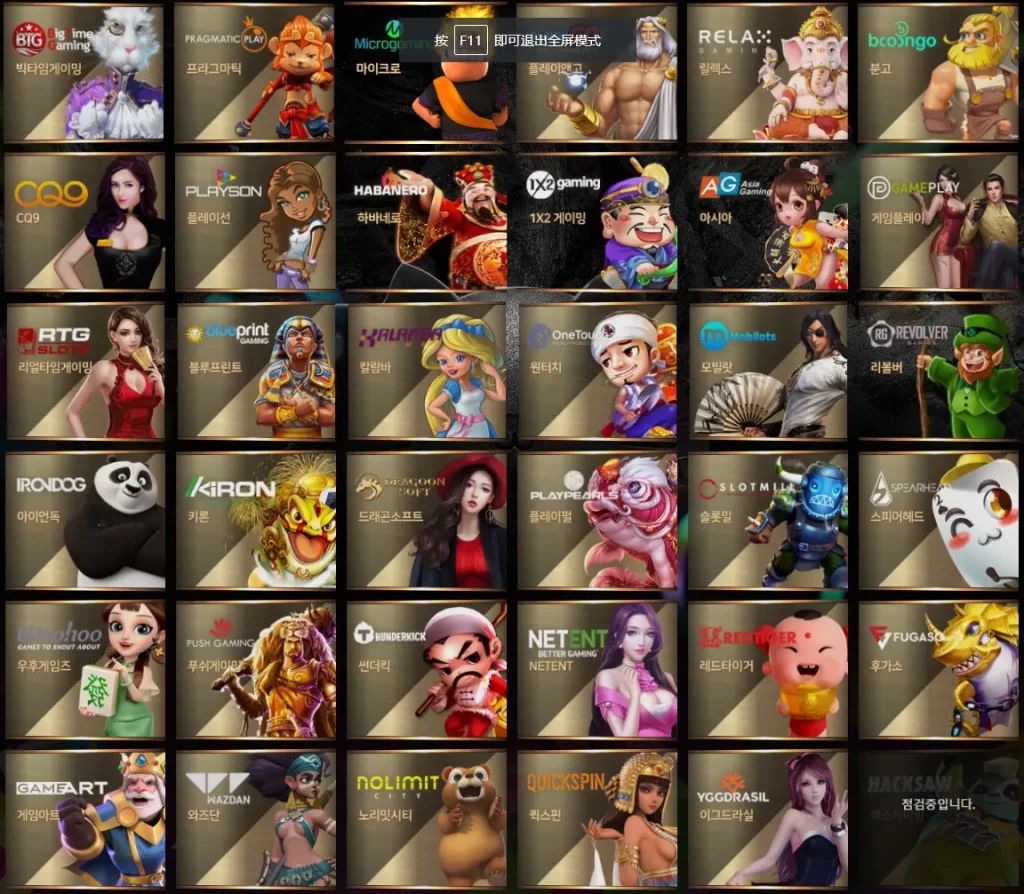
- 키론
- 플레이선
- 썬더킥
- 스피어헤드
- 슬롯밀
- 릴렉스
- 플레이펄
- 아이언독
- 모빌랏
- 플레이앤고
- 판타즈마
- 스피어헤드
- 아리스토클랏
- 마이크로
- 노리밋시티
- 릴렉스
- 마이크로
- 플레이선
- 게임피시 글로벌
- 게임아트
- 아리스토클랏
- 푸쉬게이밍
- 원터치
- 아리스토클랏
- 이그드라실
- 이엘케이
- 리얼타임게이밍
- 후가소
- 분고
- 원터치
- 리얼타임게이밍
- 원터치
- 에보플레이
- 아이언독
- 레드레이크
- 빅타임게이밍
메가슬롯 후기 이벤트

프리스핀데이 이벤트
수요일은 메가 프리스핀 데이 이벤트가 있는 날입니다
- 매주 수요일 활동내역별 프리스핀데이 쿠폰지급(해당 주 월요일, 화요일 활동내역)
- 고객센터 채팅창으로 이벤트 신청 후 이용할 게임 확인 / 본사에서 정한 슬롯게임만 이용 가능
- 프리스핀 쿠폰은 수요일 하루만 사용 가능
- 최대 출금 3배, 롤링 3배
- 프리스핀 쿠폰은 절대 입금액과 동시에 사용 금지, 사용 시 전액 몰수 처리
메가슬롯 신규가입 이벤트
메가슬롯 신규 가입 시, 가입 첫 충전에 대한 보너스가 아래와 같이 제공됩니다.
신규가입 첫충전 보너스 20% + 1만 쿠폰(최대 100만)
가입 후 1회 입금 20%, 2회 10%, 3회 5% 보너스 지급
매일 첫입금 보너스 10%(최대 100만)
매일 재입금 보너스 5%(최대 100만)
레벨별 롤링콤프
등급 / 롤링콤프 / 조건 / 첫충보너스
- 레드 / 0.1% / 가입시 적용 / 신규첫충 20% 이후 10%
- 실버 / 0.1% / 누적충전 1천만원 이상 / 첫충 10%
- 골드 / 0.2% / 누적충전 1억원 이상 / 첫충 10%
- 플래티눔 / 0.25% / 누적충전 4억원 이상 / 첫충 10%
- 에메랄드 / 0.3% / 누적충전 8억원 이상 / 첫충 11%
- 루비 / 0.4% / 누적충전 15억원 이상 / 첫충 12%
- VIP / 0.5% / 누적충전 25억원 이상 / 첫충 15%
- VVIP / 0.6% / 누적충전 60억원 이상 / 첫충 15%
첫충 보너스 최대 100만
콤프는 일요일에 만 원 이상의 금액으로 천 원 단위별로 사용 가능합니다.
메가슬롯 지인추천 이벤트
추천인 수에 따라 아래와 같이 쿠폰 지급
- 추천인 3명 – 6만원 쿠폰
- 추천인 5명 – 10만원 쿠폰
- 추천인 10명 – 20만원 쿠폰
추천인으로 한 명을 인정받으려면, 최소 세 번 입금하고 총 입금액이 50만 원 이상이어야 합니다
추천인당 0.1% 배팅롤링콤프 영구 적용!
[주의사항]
- 매주 수요일 마다 1:1 문의로 추천인 쿠폰 요청시 발급
- 여러 계정의 사용이나 같은 IP 주소에서의 접속이 확인되면, 머니는 압수됩니다
- 쿠폰만 사용 시 롤링 300%, 최대출금 3배까지 지급
- 이벤트 및 보너스 악용 회원은 게임 이용 제재
- 이벤트 기간 동안 이벤트 내용은 변경될 수 있습니다.
텔레그램 공식채널 이벤트
모든 메가슬롯 공식 채널 참가자에게 20,000원의 쿠폰이 제공됩니다!
공식 채널에 참여하여 최신 공지와 정보를 신속히 접하실 것을 추천드립니다.
[이벤트 참여 방법]
- 고객센터를 통해 공식채널 링크 문의
- 공식채널 입장 후 고객센터로 스크린샷 첨부
- 채널 이벤트 2만 쿠폰 확인

메가 카지노 : 모두의슬롯

여러분은 온라인 슬롯 게임의 세계로 스릴 넘치는 여행을 시작할 준비가 되었나요? 여러분의 인생을 바꿀 수도 있는 거대한 상품들의 약속으로 릴이 회전하고 있는 메가슬롯만 보아도 됩니다! 고품질의 그래픽, 혁신적인 기능, 그리고 풍부한 흥분이 결합된 몰입형 게임 경험에 사로잡힐 준비를 하세요. 그것은 큰 승리와 수익성 있는 보상을 찾기 위해 릴을 굴릴 때 여러분의 내적 모험가를 분출할 수 있는 기회를 제공합니다.
다양한 주제와 흥미로운 보너스 라운드로 모든 스핀은 기대와 잠재적인 부를 담고 있습니다. 클래식한 과일 슬롯을 좋아하거나 현대적인 비디오 슬롯에 매료되었다면 모든 취향을 만족시킬 게 있습니다. 이미 전 세계의 많은 플레이어들이 그 흥미를 발견했으니, 당신도 대담한 아드레날린 세계에 참여하세요.
메가슬롯을 플레이하는 것은 현재의 큰 보상을 얻을 수 있는 흥미로운 기회를 제공하는 즐거운 경험입니다. 먼저 원하는 장치에서 손쉽게 게임을 시작하세요. 목표는 릴 전체에서 일치하는 기호를 사용하여 승리 조합을 만드는 것입니다. 이를 위해 게임의 작동 방식을 이해해야 할 것입니다. 일반적으로 다섯 개의 릴과 여러 개의 페이라인 또는 승리 방법으로 구성됩니다.
메가슬롯 주소 | 슬롯 가입쿠폰, 지금 당신의 것으로 얻으세요!

릴을 회전하기 전에 베팅 크기를 기호에 따라 조절합니다. 그런 다음 스핀 버튼을 누르고 기호가 무작위로 나타나는 것을 확인합니다. 와일드나 산점과 같은 특별한 기호를 주시하고, 자유 스핀이나 미니 게임과 같은 흥미로운 보너스 기능을 활성화할 수 있습니다. 일부 메가슬롯은 무작위로 또는 특정 기호 조합으로 얻을 수 있는 점진적인 잭팟도 제공합니다.
메가슬롯의 세계에서 플레이어들은 릴이 돌아가는 흥미 뿐만 아니라 엄청난 보상의 약속으로 인생을 현실로 바꿀 수 있는 기회를 얻습니다. 캐시 잭팟부터 이국적인 여행지에서의 호화로운 휴가까지 다양한 수익성 있는 보상이 플레이어들을 기다립니다.
초보자든 숙련된 플레이어든 간에, 항상 놀라운 것이 있습니다. 하지만 그것은 단지 큰 승리에 관한 것이 아닙니다 – 그것은 모든 플레이어가 보상을 받을 수 있는 기회를 보장합니다. 다양한 보너스 기능과 게임 플레이에 포함된 미니 게임으로, 더 작은 승리도 합쳐지고 아주 신나는 경험을 만들 수 있습니다. 그러므로, 다른 어떤 것들과 같은 모험을 준비하세요.
투데이슬롯

게임의 부드러운 운용은 사용자가 액션에 깊이 몰입하게 만드는 매혹적인 경험을 제공합니다. 눈길을 끄는 비주얼과 매력적인 애니메이션은 게임의 각 순간을 생동감 있게 만들어 전체적인 플레이 품질을 높여줍니다. 다양한 보너스 라운드와 무료 스핀 기능을 통해 큰 이익을 얻을 수 있는 기회가 늘어납니다.
메가슬롯 게임은 다양한 특별 기능과 보너스 라운드를 제공하여 플레이어에게 스릴 넘치는 게임 경험을 선사하며, 이를 통해 승리할 기회를 더 많이 제공합니다. 와일드 기호는 눈에 띄는 기능으로, 다른 기호들을 대체할 수 있는 뚜렷한 로고로 표현되어, 승리 조합을 이룰 가능성을 향상시킵니다. 또한, 스캐터 기호는 게임을 한층 더 흥미롭게 만드는 보너스 라운드를 활성화시킬 수 있습니다.
메가슬롯 게임에 포함된 프리 스핀 기능은 스릴 넘치는 추가적인 특징으로, 스캐터 기호 3개를 모으면 무료로 스핀할 수 있는 기회가 주어지고, 해당 기간 동안 이루어지는 모든 승리는 배가 됩니다. 이는 추가적인 비용 부담 없이 큰 이득을 얻을 수 있는 좋은 기회며, 또한 보너스 라운드에서는 독창적인 미니 게임이 등장합니다. 이 게임들은 플레이어의 승리 가능성을 최대화하기 위해 전략적 사고를 필요로 합니다.
메가슬롯 조작 FAQ 자주 묻는 질문
메가슬롯에서 다른 카지노나 스포츠 베팅도 지원하나요?
아니오, 메가슬롯은 전적으로 슬롯 게임만을 다루는 전용 사이트로, 오직 정품 게임만을 제공합니다. 이로 인해 슬롯 게임의 질과 양 모두에서 최고의 퀄리티를 제공할 수 있습니다.
메가슬롯 궁금한 점이나 문제가 생겼을 때 어디에 문의하면 되나요?
24시간 동안 공식 사이트 하단에 제공된 고객센터 계정으로 문의하시면 즉각적인 답변을 제공합니다. 텔레그램 공식 채널 가입 이벤트의 혜택도 살펴보시어 이용하시길 바랍니다.

